Preface
“If you are headed in the right direction, you will inevitably encounter and solve the right problems.” – Michael Sherer
Looking at health care in the USA, it is hard not to conclude that we are headed in the wrong direction. Our healthcare system, independent of any critique we might have of our approach to health insurance:
- manages diseases rather than preventing and curing them.
- classifies too many diseases as incurable and of unknown origin.
- intervenes too late in the disease process.
- is over-reliant on prescription drugs and surgical procedures
- is incredibly costly. US healthcare spending is approaching 20% of GDP. That’s not sustainable even for the wealthiest country on earth.
- is too far behind the research. We’ve learned so much about how the body works in the past decade and it really hasn’t affected how we prevent and treat disease.
- Is being undermined by politics and profit motive.
Current political efforts to improve things have devolved into a partisan foodfight over whose wrong answer is best.
While it would be tempting to despair of the situation ever getting better, I believe the seeds of renewal and reformation of our approach to health care have already been sown in the global medical research community. Over the last decade or so, scientists in the fields of redox biology, biochemistry, epigenetics, various specializations ending in -omics (ie. proteomics, metabolomics, transcriptomics, etc.) and specific disease research have been systematically detailing how the cell works, revealing the causes of chronic disease and the mechanisms by which it might be prevented and cured.
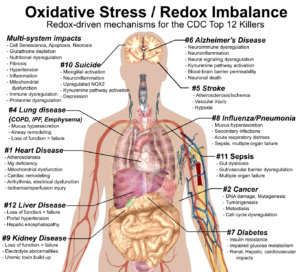
At the heart of this scientific sleuthing tale is redox imbalance, a condition where chronic stress causes elevated oxidative processes (oxidative stress), which diminishes antioxidative capacity (the body’s ability to defend itself), making the body vulnerable to the cellular and systemic damage and dysregulation that leads to chronic diseases like aging, cancer, heart disease, diabetes, lung diseases, neurodegenerative diseases, autoimmune disease, etc.
Surprisingly, this body of work is largely unknown even though it is quite extensive–over 230,000 articles on oxidative stress on PubMed at this writing–and growing at the rate of nearly 50 new articles per day. Perhaps it is because the underlying biochemical processes are complex and defy simple explanations, that the treatments are largely non-pharmaceutical, or the testing methodologies are unstandardized and unfamiliar to doctors. Whatever the reason, I believe the story of redox imbalance represents “the right direction” we need to head. In so doing, we will encounter the right problems:
- How to screen for redox imbalance–frequently, reliably, inexpensively
- What biomarkers to follow and test for
- What the most effective treatment protocols are for prevention
- How to reverse oxidative damage and dysregulation once it has occurred.
- And more….
And we will solve them.
The 95 theses that follow are a bit of teaser–statements that reflect the state of the research at this writing, and could be taken at face value, but could also have lots of additional detail, even entire chapters devoted to their exploration.
95 Theses for a Healthcare Reformation
In 1517 Martin Luther launched the Protestant Reformation by nailing his 95 Theses to the Wittenburg Church door. Five hundred years later, let these 95 theses concerning redox imbalance spark a much-needed Healthcare Reformation:
- Over $1.3 trillion in annual recurring healthcare costs in the US alone go to treat diseases that are caused by a condition that western medicine does not currently measure, diagnose or treat.
- That condition is redox imbalance, where elevated oxidative stress and diminished antioxidant capacity cause a chronic, pro-oxidant state, or stated more generally, oxidative processes are operating outside the normal range of redox steady state or homeostasis.
- Redox (oxidation/reduction) reactions are pervasive and necessary to sustain life, yet dangerous if dysregulated. Redox reactions are foundational to cellular respiration, signaling, and immune function, yet unless tightly-regulated, reactive oxygen and nitrogen species (ROS/RNS) will damage, dysregulate or destroy cells and tissue, dysregulate key bodily functions, generate toxic compounds, and catalyze inflammatory cascades that cause systemic inflammation, dysregulation and disease.
- Redox imbalance is a root cause of most chronic disease and even most acute disease. Those diseases include cardiovascular disease, cancers, diabetes, chronic lung disease, chronic liver disease, autoimmune disease, chronic kidney disease, neuropsychiatric and neurodegenerative diseases, skin diseases, rheumatic diseases, digestive diseases, thyroid diseases, musculoskeletal diseases and aging.
- Your body is designed to handle and even benefit from normal levels of oxidant (ROS/RNS) production thanks to its endogenous (internal) antioxidant system. This system, anchored by NRF2, glutathione, superoxide dismutase, catalase and related enzymes, is designed to keep oxidation in check and raise antioxidant defenses when stimulated by stressors.
- Your body is NOT designed to handle chronic stress. Over time, elevated stress inputs can dysregulate your body’s antioxidant system, depleting reduced glutathione levels (GSH), NAD+ and related antioxidant nutrients (ie. selenium, zinc, magnesium, B vitamins, amino acids), and lowering antioxidant capacity.
- A body with a depleted antioxidant system is vulnerable to future stress, damage, dysregulation and disease. When antioxidant capacity is low, the body cannot mount an effective antioxidant response to even a minor oxidative stressor, making it more vulnerable to damage and dysregulation.
- Stress has many forms and your body’s cells respond to them all by producing Reactive Oxygen and Nitrogen Species (ROS/RNS)–a Universal Stress Response. While we are most familiar with psychological stress, oxidative stressors also include pathogenic stressors (viruses, bacteria, fungi), physical stress (exercise, injury, misalignment), toxic stress (cellular waste products, oxidation byproducts, xenobiotics, heavy metals), environmental stress (heat, light, sound, smells, weather changes, etc.) and nutritional stress ( overconsumption, highly processed foods, high sugar diets, low fruit and vegetable intake and resulting vitamin and mineral deficits). ROS and RNS perform useful, necessary signaling functions, but damaged lipids, proteins, carbohydrates, DNA and cause pathological signaling at high levels.
- Elevated levels of circulating hydrogen peroxide (H2O2) cause pathological signaling that leads to disease. This situation occurs in redox imbalance, when diminished antioxidant capacity fails to reduce H2O2 radicals into water and O2.
- Though less studied, there is a phenomenon called reductive stress where overuse of antioxidants suppress the beneficial effects of normal ROS production and/or cause pathological signaling and dysregulation.
- The stress response progresses from Alarm > Resistance > Fatigue. Alarm is characterized by the release of stress hormones to mobilize the bodies short-term stress response. Resistance is characterized by short-term increase in antioxidant capacity, immune system activation, while fatigue is characterized by diminishing antioxidant capacity, and increasingly incompetent or dysregulated functioning of protective systems (ie. the immune system, the nervous system, the HPA Axis).
- Researchers know how to measure oxidative stress. Current methods include measuring blood levels of oxidative by-products such as malondialdehyde (MDA) and isoprostanes, lipid oxidation byproducts, advanced oxidized protein products (AOPP’s), thiobarbituric acid reacting substance (TBARS) and Advanced Glycation Endproducts. Other biomarkers are under investigation.
- Researchers know how to measure antioxidant capacity. The GSH/GSSG ratio (reduced glutathione / oxidized glutathione) is indicative of total antioxidant capacity. Glutathione peroxidase 1 (GPX-1) has also shown promise as a marker of serum antioxidant capacity.
- Researchers know how to raise antioxidant capacity. Studies have shown that supplementing with 2 – 2.4 g/day of n-acetylcysteine reliably raises glutathione levels in humans. This makes n-acetylcysteine a common reference tool in research studies on the role of redox imbalance in disease states. Magnesium, Vitamin B12, Vitamin D3 and plant polyphenols have all shown the ability to raise antioxidant capacity.
- The NRF2 transcription factor is a key regulator of antioxidant activity and cellular defense. Upregulating NRF2 with plant polyphenols, exercise, fasting and ketogenic diets raises antioxidant capacity.
- Raising antioxidant capacity, when low, has broad therapeutic benefit. Studies using N-acetylcysteine supplementation to raise glutathione levels/antioxidant capacity have shown clinical improvement in Lupus symptoms, prevention of influenza and shortened illness duration, protection of the liver and kidney from oxidative damage, lowered homocysteine levels, improved peripheral neuropathy, and generally prevention of oxidative damage from a wide range of toxic and oxidative insults. Studies on plant polyphenols have repeatedly shown that they protect the body from oxidative damage and dysregulation by raising antioxidant capacity.
- Nutritional status plays a key role in maintaining healthy redox status. Other compounds that play a role in maintaining healthy redox status include glutathione precursors L-glutamine, glycine, and associated redox co-factors vitamin C, vitamin E, vitamins B1, B2, B6, B12, folate (B9), minerals magnesium, selenium and zinc, and alpha lipoic acid.
- Human beings are designed to eat plants, some meat, but not large quantities of sugar and processed foods.
- The Western Diet, with its dependence on over-consumption of high-calorie, low-nutrient, processed foods, is a chronic contributor to oxidative stress and redox imbalance.
- A nutrient-dense diet rich in fruits and vegetables, leafy greens, legumes, nuts and seeds helps maintain healthy redox balance.
- A healthy gut ecosystem helps maintain healthy redox balance. Fiber and resistance starch increase production of short chain fatty acids in the gut. Butyrate in particular modulates oxidative stress in the colonic mucosa, protecting against colon cancer.
- Amino acids are key players in maintaining redox balance, both as precursors of glutathione (cysteine, glutamine, glycine) and facilitators of redox-related reactions (tyrosine, arginine, creatine, cystine, isoleucine, lysine, methionine, proline, phenylalanine, etc.)
- Sirtuins, a family of 7 proteins, play a regulatory role in redox balance, cardioprotection and neuroprotection. SIRT1 is upregulated by calorie restriction, resveratrol and exercise and protects against oxidative damage, slows aging and protects against diabetes, liver and kidney damage, UV skin damage, etc. SIRT3 protects against tumorigenesis. SIRT6 is cardioprotective.
- Oxidative stress activates the immune system via the NFkB pathway
- People with low antioxidant capacity have dysregulated, typically over-reactive immune systems.
- The immune system uses oxidative stress as a weapon to kill pathogens and neutralize toxins. Macrophages, neutrophils, monocytes and phagocytes employ what is called an oxidative burst to kill pathogens.
- Activation of the immune complement system can cause a major increase in oxidative stress, inflammation and disease symptoms. People who are highly reactive to a broad range of foods and chemicals may want to avoid dietary lectins, which can activate the complement system via the lectin complement pathway.
- Chronic infection (bacterial, viral, fungal), often undetected, is a key contributor redox imbalance. Our current inability to reliably detect and eradicate these infections contributes to pathogenesis and progression of oxidative stress-driven diseases.
- Redox imbalance is a root cause of allergies, asthma and autoimmune response because it invokes the immune system for minor oxidative stressors that would otherwise be dealt with by your body’s antioxidant system.
- Redox imbalance causes the body’s immune system to be chronically activated, which leads to systemic inflammation, immune hyper-reactivity and immuno-incompetence.
- Lipopolysaccharide is a key player systemic inflammation and redox imbalance. Chronically impaired gut barrier function and increased permeability of blood vessels contribute to redox imbalance by allowing lipopolysaccharide, a toxin found in the cell walls of bacteria, to pass into the bloodstream causing a condition called endotoxemia and resulting systemic oxidative stress, inflammation, neuroinflammation and tissue damage.
- When antioxidant capacity is low, even mild oxidative stressors can push the body into an inflammatory state. Examples would include weather-change migraines, joint pain from eating wheat or dairy, or hives from an allergen.
- Oxidative stress dysregulates nitrogen metabolism.
- Oxidative stress causes the production of Reactive Nitrogen Species (RNS) such as Peroxynitrite, a key pathogenic mechanism in cardiovascular disease, diabetes, neurodegenerative diseases and cancer.
- Peroxynitrite is formed from Nitric Oxide (NO) and a superoxide (O2) radical.
- NADPH oxidase and Xanthine oxidase can produce large amounts of O2. So when these enzymes are elevated, peroxynitrite formation can increase exponentially, causing inflammation and cellular/tissue damage.
- Plant polyphenols like quercetin downregulate NADPH oxidase and Xanthine Oxidase.
- Inhibition of the enzyme Arginase is a key strategy for regulating nitrosative stress, since arginase competes with the enzyme endothelial constitutive nitric oxide synthase (eNOS) for arginine and thus prevents NO production. Ginger, curumin and quercetin are natural compounds that downregulate arginase.
- Exogenous antioxidants (ie. plant phytonutrients and antioxidant supplements) are beneficial in moderation and potentially damaging at high doses. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2952083/
- Exercise increases glutathione levels through ROS signaling and resulting increase in glutathione reductase activity.
- Processed carbohydrates and sugar are oxidative stressors
- Overeating is an oxidative stressor.
- Alcohol contributes to ROS generation and interferes with the antioxidant response. http://pubs.niaaa.nih.gov/publications/arh27-4/277-284.htm
- Redox Imbalance causes Signaling Dysregulation. This impacts multiple systems including the Sympathetic Nervous System, the HPA Axis, the immune system, the Renin Angiotensin Aldosterone system, and cellular processes like autophagy, angiogenesis, apoptosis, mitochondrial energy production and protein folding.
- Redox Imbalance causes Genetic/Epigenetic Dysregulation via direct DNA and RNA damage and altered signaling pathways.
- Redox Imbalance causes Detoxification Dysregulation by depleting glutathione levels and diminished liver and kidney function.
- Redox Imbalance causes Nutritional Dysregulation. Oxidative stress and in turn, reduced nutritional status impairs the body’s ability to mount an effective antioxidant response to an oxidative stressor.
- Oxidative stress causes intracellular magnesium depletion. Low intracellular magnesium levels are associated with joint and tendon problems, muscle cramps, twitches and tension, electrical issues in the heart (ie. tachycardia), atherosclerosis, heart failure, sudden cardiac death, skin diseases, calcium dysregulation, low vitamin D levels, impaired antioxidant response, etc.
- Oxidative stress disrupts cellular calcium homeostasis and should be considered an early impact of redox imbalance. Elevated cellular ca2 levels increase oxidative stress and can lead to cell death. `
- Redox Imbalance causes metabolic dysregulation, often referred to as metabolic syndrome.
- Oxidative stress damages cell mitochondria, leading to mitochondrial dysfunction and metabolic syndrome.
- Mitochondrial damage and dysfunction should be considered an early and ongoing effect of redox imbalance, with significant increased risk for chronic disease.
- Mitochondrial dysfunction is associated with aging, neurodegenerative diseases, cancer, diabetes and heart disease.
- Oxidative stress causes endoplasmic reticulum stress and dysfunction.
- Endoplasmic Reticulum stress caused by oxidative stress is a major disease mechanism. The endoplasmic reticulum (ER) play key regulatory roles in the cell through intracellular calcium regulation and their protein synthesis and protein folding functions. Dysregulated ER play a pathogenic role in elevated cellular oxidative stress, cellular calcium dysregulation and accumulation of unfolded/misfolded proteins. This contributes to cell dysfunction and death. ER stress also plays a role in the progression of neurodegenerative and cardiovascular diseases.
- Redox Imbalance causes ecological dysregulation in the form of gut dysbiosis and dysregulation of other microbiome locations.
- Elevated oxidative stress plays a central role in inflammatory bowel diseases, via a dysregulated gut microbiome and activation of the sympathetic nervous system.
- Redox imbalance activates your sympathetic nervous system (fight or flight mode), which prevents your parasympathetic nervous system (rest and repair mode) from doing its health-restoring work.
- When your nervous system is in sympathetic (fight or flight) mode, energy is directed away from digestion, reproduction and repair processes. Your digestion slows down so that all energy can be directed towards addressing the immediate threat (ie. escaping the bear). This helps explain the role of oxidative stress in issues like gastroparesis and acid reflux (GERD).
- Redox imbalance causes the release of catabolic hormones like cortisol, which leads to central obesity.
- Oxidative stress depletes arginine by stimulating arginase activity. Arginine is a Nitric Oxide (NO) donor, so its depletion results reduced NO levels in digestive tissues and reduced gastric motility. See http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3268202/
- Low NO levels in vascular tissue contributes to arterial stiffness, constriction, poor circulation and high blood pressure.
- Smoking is a major oxidative stressor on the lungs and beyond. The negative health impacts of smoking (impaired lung function, increased infection levels, DNA damage, cancer, heart disease, stroke, diabetes, periodontal disease, reproductive issues, etc.) can largely be understood in terms of its chronic oxidative stress burden. While tobacco is the best understood, all forms of smoking (marijuana, e-cigarettes, hookah) are oxidative stressors with the potential for negative health consequences.
- Oxidative stress plays a key role in neuropsychiatric illnesses including bipolar depression, major depression, schizophrenia, ADHD, OCD. The brain is particularly ill-equipped to handle elevated oxidative stress levels, so systemic elevation of oxidative stress will cause neuroinflammation and dysregulation of neurological systems and processes.
- Oxidative stress is implicated in the development of neurodegenerative diseases including Parkinson’s Disease, Huntington’s disease, Alzheimer’s Disease, and ALS.
- Oxidative stress plays a central role in male and female infertility. Supplementing with n-acetylcysteine and other antioxidants improves both male and female fertility.
- Oxidative stress plays a contributing role to birth defects and adverse pregnancy outcomes, such as stillbirth. It is not unreasonable to assume that higher rates of birth defects seen in Zika infection, diabetes and smoking are related to elevated oxidative stress levels associated with each of these conditions.
- Lack of sleep and poor sleep quality are significant causes of oxidative stress.
- Melatonin, the sleep hormone, is a powerful antioxidant that protects the brain from oxidative damage and dysregulation. Melatonin production is cyclically tied to the lunar phases, and low at full moon. This in part explains behavioral issues at full moon at public schools, nursing homes and mental hospitals.
- Elevated oxidative stress is central to the pathologies in genetic conditions like Down Syndrome and FragileX syndrome.
- Elevated oxidative stress and lowered magnesium status is associated with hearing loss.
- Elevated oxidative stress and diminished antioxidant capacity is associated with macular degeneration, diabetic retinopathy, glaucoma, cataract and other eye diseases.
- Workers with healthy redox balance take fewer sick days. http://jn.nutrition.org/content/137/12/2737.long
- People with healthy redox balance have fewer colds, sinus infections and upper respiratory infections.
- People with health redox balance are resistant to autoimmune and inflammatory diseases.
- People with healthy redox balance are less likely to get cancer
- People with healthy redox balance are less likely to get diabetes
- People with healthy redox balance are less likely to be depressed
- People with healthy redox balance are less likely to develop heart disease.
- People with healthy redox balance are less likely to develop chronic liver and kidney disease.
- Infants with healthy redox balance and nutritional status are less prone to sudden infant death syndrome.
- Children with healthy redox balance are less likely to develop autism
- Children with healthy redox balance are less likely to have ADHD and other neuropsychiatric issues.
- Oxidative stress has long been linked the pathologies of aging (see the antioxidant theory of aging).
- Glutathione and NAD+ levels decline with age. This means reduced resilience to stressors (low glutathione) and reduced energy production and Sirtuin activity (low NAD+ levels).
- In aging, oxidative stress causes cells to enter a state called senescence, where they no longer reproduce. Senescence is a pathophysiological mechanism in diseases like pulmonary fibrosis.
- Elderly people with healthy redox balance and nutritional status are less likely to have dementia and neurodegenerative diseases, sarcopenia, osteoporosis and other musculoskeletal ailments.
- For diagnosis of redox imbalance, while biomarkers will continue to evolve, GSH/GSSG ratio (reduced glutathione/glutathione disulfide) is a reasonable biomarker for antioxidant capacity, while TBARS and MDA (malondialdehyde) are useful biomarkers for spotting elevated oxidative stress. Beyond that, testing for serum lipopolysaccharide levels (a significant source of oxidative stress) will help you understand whether there is intestinal permeability / bacterial translocation and probably gut dysbiosis.
- Therapeutic strategies for redox imbalance are largely non-pharmaceutical and should focus on identifying and minimizing oxidative stress inputs, restoring endogenous antioxidant capacity (glutathione, superoxide dismutase, catalase and related enzymes) and restoring nutritional status. Targeted use of polyphenols, other exogenous antioxidants, amino acids, vitamins, minerals and omega-3 fatty acids as medical interventions can be useful.
- NRF2 and NAD+ are emerging therapeutic targets for maintaining and restoring healthy redox balance and mitochondrial function.
- Health practitioners and consumers will have a learning curve with the new redox health paradigm, but to the extent that it is a more accurate portrayal of the disease process and leads to better outcomes, it will be a welcome change.
- By NOT measuring, diagnosing and treating redox imbalance and associated disorders, mainstream medicine is missing a key opportunity to prevent or delay the onset of chronic disease. They are also missing an opportunity to improve the outcomes of existing treatment protocols.
- By NOT measuring, diagnosing and treating redox imbalance we perpetuate a medical system that manages diseases rather than cures them, treats symptoms rather than root causes, intervenes too late to prevent damage and dysregulation, is over-reliant on pharmaceuticals that mask symptoms, often contributing to further dysregulation–all resulting in care that more costly and less effective that it needs to be.
- MEDICAL SCIENCE HAS CREATED AN OVERWHELMING CASE FOR THE CENTRALITY OF OXIDATIVE STRESS TO THE PATHOGENESIS AND PROGRESSION OF CHRONIC DISEASE.
- IT IS TIME FOR THE PRACTITIONERS OF WESTERN MEDICINE TO MEASURE, DIAGNOSIS AND TREAT REDOX IMBALANCE.
Postscript
Summarizing the 95 Theses for Healthcare Consumers
How do you take something complex and make it understandable and useful for everyone? It is somewhat paradoxical that I am presenting highly-technical jargon in the 95 theses in hopes that both medical professionals and ordinary healthcare consumers will be convinced to view health and disease differently–adopting a different health and disease paradigm.
My optimism that this might be possible stems from the reality that, despite the incredible complexity of the human body, I am proposing something simple: that chronic disease stems from the imbalance of a single, universal cellular process–the regulation of oxidation and reduction (redox) processes–caused by chronic stress in all its forms. If you can restore that (redox) balance, your body’s own health-seeking homeostatic processes will do their best to restore your sense of health and well-being.
I find that most people intuitively find this to be plausible. How many times have you said things like ‘I got this cold because things are really stressful at work and my immune system is run down’ or ‘I was under a lot of stress and came down with shingles (or some other condition).’ We recognize that stress makes us vulnerable to disease. Why should it surprise us that there is a biochemical explanation for why this is so?
Like any hypothesis, the idea that there is one root cause, one disease–redox imbalance, deserves to be tested. Does it hold up? Does it explain the many manifestations of health and disease better than the current medical paradigm? If I apply the underlying principles behind redox imbalance and how the body breaks down to some kind of health intervention, do I get better? In my experience, the answer to all these questions is yes. While a detailed explanation of the science behind the redox health paradigm is beyond the scope of this blog post, I will leave you with a short list of learnings and ramifications growing out of this approach that I believe are applicable for a broad range of health consumers:
- Fruits and vegetables contain polyphenols and other micronutrients that are important regulators of redox balance. This in large part explains why whole-food plant-based diets are increasingly recognized as health-promoting.
- Processed carbohydrates, sugars, artificial sweeteners, grain oils, meat (particularly beef) alcohol and smoking are all oxidative stressors, and this significantly explains why they degrade health over time.
- Lack of sleep and poor sleep quality are stressors and over time will impact health.
- Stress and oxidative stress activate the immune system and over time degrade its function
- Chronic stress ‘uses up’ antioxidant compounds which then need to be replenished to restore healthy redox balance. Among these are magnesium, zinc, selenium, Vitamin B12, Vitamin D3 and reduced glutathione.
- Infections–bacterial, viral and fungal–are important sources of oxidative stress. Low-grade chronic infections are an important and under-recognized source of chronic disease because they keep the immune system activated and under-performing and cause inflammation that manifests itself as joint pain, headaches, etc.
- There is a very strong association (most likely causal) between oxidative stress and magnesium deficiency and this association plays a much greater role in cardiovascular disease than is currently recognized. Because of magnesium’s role in the electrical function of the heart, this relationship between oxidative stress and magnesium deficiency explains why people under stress experience sudden cardiac death, atrial fibrillation and PVC’s. It also suggests that magnesium supplementation is probably one of the lowest cost strategies for preventing heart disease.
- Plant polyphenols are non-patented wonder drugs, in that they have multiple health benefits and relatively few side effects unless you take excessive amounts. The most popular include quercetin, rutin, resveratrol, pterostilbene, astaxanthin, hesperidin, curcumin, ECGC, pterostilbene and resveratrol. Polyphenols can generally be thought of as mild eustressors that stimulate NRF2 to raise antioxidant protection levels in the cell.
- While the pharmaceutical industry warns of a shortage of antibiotics, there are natural compounds that fight not only bacteria, but also viruses and fungi and do so in much less disruptive ways than standard antibiotics. Examples would include oil of oregano, olive leaf extract, garlic oil, bee propolis, n-acetylcysteine and lactoferrin. Keeping these on hand for fighting off colds and respiratory infection is enormously useful
- Glutathione, your body’s primary antioxidant also plays an important role in immune function and detoxification. Your liver is a glutathione exporter.
- Exercise is a stressor that demonstrates the concept of hormesis, where oxidative stress stimulates the body’s antioxidant capacity to become more resilient. It also explains why over-training has negative health consequences, such as increased upper respiratory infections.
- Fasting is a stressor that like exercise, bolsters resilience and is associated with longevity.
- Early impacts of oxidative stress include immune system activation, raising of antioxidant defenses, activation of your adrenal system, activation of your sympathetic nervous system and calcium loading in the cell. This is your body on full alert, preparing to defend itself.
- As your body defends itself from chronic stressors, antioxidant systems are progressively depleted, energy production is impaired (mitochondrial dysfunction), cells and tissues are damaged, toxic compounds are created, inflammation increases, blood vessel permeability increases and bodily systems become increasingly dysregulated, damaged and fatigued. This can manifest itself as fatigue, pain, loss of mental acuity, rashes, depression, anxiety, hyperactivity, hypersensitivity, bone loss, muscle loss or even tumors, nerve damage and organ damage.
- We know what death by oxidative stress looks like. It’s called sepsis and it’s what happens when the body is overwhelmed by a bacterial infection and associated toxins (ie. lipopolysaccharide) and ultimately organ systems shut down resulting in death.
- Scientists have long theorized that oxidative stress is the root cause of aging. In line with that hypothesis, glutathione levels and NAD+ levels fall with age, resulting in lower energy production, weaker immune response, and lowered resistance to stressors.
- Permeability of the intestinal barrier, blood-brain barrier and blood vessels general is an under-recognized cause of inflammation, infection, oxidative stress and ultimately disease, as bacteria and toxic compounds from the gut pass into the blood stream and must be dealt with by the immune system.
- Mindfulness, meditation and other mental stress management techniques are important because humans have the unique ability to generate stress responses simply by thinking about things that might happen. Meditation and prayer can reduce sympathetic nervous system activation.
- Intervening as close to the root cause as possible will yield better outcomes for patients and their doctors..
As we look to the future, it’s not hard to imagine a reality where testing for a range of redox biomarkers is continuous and cheap or even free. We’ll spot imbalances as they occur and receive personalized recommendations for how to rectify them. The interventions could be largely non-pharmaceutical. The personalized recommendations could be recipes or lifestyle changes rather than drugs. Healthcare costs would go down and health status would improve. We could solve more of our own health issues and reserve our doctor visits for true health crises. We could live well into old age and die at home. That is the reformation I am seeking after.
Best wishes,
Michael Sherer